Image gallery for: Diels alder reaction
- Advertisement
-

In these practice problems, we will discuss the endo and exo products of the Diels–Alder reaction addressing the regiochemistry and stereochemistry
-

As requested, here are some organic chemistry notes from a basic organic chemistry class that I took in spring 2011. The order of these notes are not the order in which they were learned so the inf…
-

In these practice problems, we will determien the product of Diels-Alder reactions based on the regioselectivitve reaction of the Diene and Dionophile.
-

When non-symmetrical dienes react with non-symmetrical dienophiles in the Diels Alder reaction, two regioisomers are possible. Let's look at some examples.
- Advertisement
-

Definition of pericyclic reactions What is cyclo addition reaction ? Woodward-Hoffmann selection rules for cyclo addition reaction are as follows, Exo-cyclo addition reaction Endo cyclo addition reaction What is Diels Alder reaction ? ( 4+4) type cyclo addition reaction:
-

When non-symmetrical dienes react with non-symmetrical dienophiles in the Diels Alder reaction, two regioisomers are possible. Let's look at some examples.
-
![[17] Diels-Alder Cycloaddition 1928](https://i.pinimg.com/originals/65/0b/62/650b62c40d6b339a5407284fe39b7715.png)
Visit the post for more.
-

In these practice problems, we will discuss the endo and exo products of the Diels–Alder reaction addressing the regiochemistry and stereochemistry
-

In these practice problems, we will discuss the endo and exo products of the Diels–Alder reaction addressing the regiochemistry and stereochemistry
-

70 Jahre Forschung an der Diels-Alder-Reaktion: Dieses Buch fasst die wichtigsten und beeindruckendsten Ergebnisse in einzigartiger Weise zusammen! Zunächst werden die Grundprinzipien der Reaktion klar und verständlich anhand übersichtlicher Graphiken erläutert. Spezielle Vorschriften und gegebenenfalls ihre industrielle Umsetzung werden anschließend erklärt. Einen Schwerpunkt bilden auch physikalische und katalytische Verfahren zur Steigerung der Selektivität der Reaktion. Cycloadditionen in konventionellen und unkonventionellen Medien werden vorgestellt. Mit über 1.000 Literaturverweisen!
-

The Diels Alder Reaction: Introduction, Mechanism, Key Pattern of bond-forming/breaking, Why is s-cis conformation required? + many examples and appendix
- Advertisement
- Advertisement
- Advertisement
-

In these practice problems, we will discuss the endo and exo products of the Diels–Alder reaction addressing the regiochemistry and stereochemistry
-

The "retro-Diels Alder" reaction is the exact reverse of the Diels-Alder and passes through the same transition state. Let's look at some classic examples.
-

Diels Alder tutorial video breaking down the reaction, mechanism and a trick for quickly identifying the product from reactants and starting molecules from a given product
-
![[17] Diels-Alder Cycloaddition 1928](https://i.pinimg.com/originals/7a/31/0f/7a310f7f733a0861cc8f5f7456aff83b.jpg)
Visit the post for more.
-

In these practice problems, we will discuss the endo and exo products of the Diels–Alder reaction addressing the regiochemistry and stereochemistry
-

Diels-Alder Reaction. Diels-Alder reaction: A cycloaddition reaction of a conjugated diene and certain types of double and triple bonds. dienophile: Diene-loving. Diels-Alder adduct: The product of a Diels-Alder reaction. Diels-Alder Reaction. Alkynes also function as dienophiles.
-

Sold by Create your own from scratch Style: Basic Dark T-Shirt Comfortable, casual and loose fitting, our heavyweight dark color t-shirt will quickly become one of your favorites. Made from 100% cotton, it's unisex and wears well on anyone and everyone. We’ve double-needle stitched the bottom and sleeve hems for extra durability. Select a design from our marketplace or customize it to make it uniquely yours! Size & Fit Model is 6’2” and is wearing a medium Standard fit Garment is unisex sizing Fits true to size Fabric & Care 100% cotton (Heathers are a cotton/poly blend) Double-needle hemmed sleeves and bottom Imported Machine wash cold
-

When non-symmetrical dienes react with non-symmetrical dienophiles in the Diels Alder reaction, two regioisomers are possible. Let's look at some examples.
-

In these practice problems, we will determien the product of Diels-Alder reactions based on the regioselectivitve reaction of the Diene and Dionophile.
-

How do you tell the exo and endo products apart in the Diels-Alder reaction? Here's a simple rule along with several examples to help you.
-
![[17] Diels-Alder Cycloaddition 1928](https://i.pinimg.com/originals/c2/b5/1d/c2b51d2091b73ed404013ac84afca96e.jpg)
Visit the post for more.
-

Diels-Alder reaction practic problems: Predict th emajor product of each Diels-Alder reaction addressing the stereochemistry of the reaction.
-

The Diels Alder Reaction: Introduction, Mechanism, Key Pattern of bond-forming/breaking, Why is s-cis conformation required? + many examples and appendix
-

In these practice problems, we will discuss the endo and exo products of the Diels–Alder reaction addressing the regiochemistry and stereochemistry
-

The Diels Alder Reaction: Introduction, Mechanism, Key Pattern of bond-forming/breaking, Why is s-cis conformation required? + many examples and appendix
-
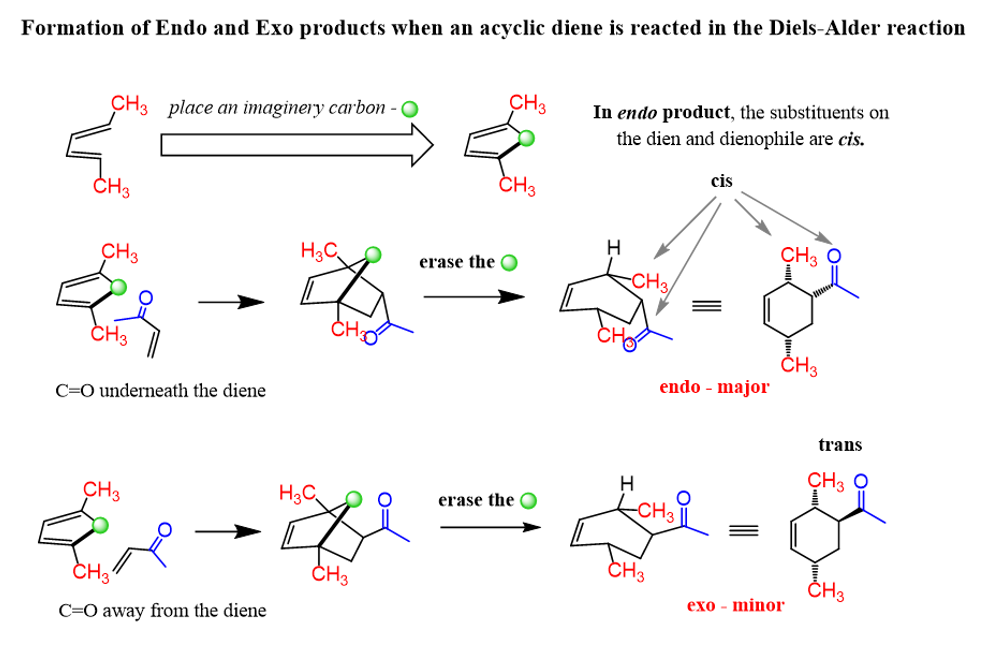
In these practice problems, we will discuss the endo and exo products of the Diels–Alder reaction addressing the regiochemistry and stereochemistry
-

(Phys.org)—The Diels-Alder reaction is a mainstay in organic chemistry. The reaction traditionally involves a diene and a dienophile. The diene has four carbons that are sp2 hybridized to form pi bonds. The dienophile has two sp2 hybridized carbons. The diene and dienophile undergo a cycloaddition reaction to form a cyclohexene. This reaction can be tweaked to make six-membered rings with a higher oxidation state by removing hydrogens from the reactants to make sp hybridized carbons. For example, rather than an alkene and a diene reacting, the reactants could be an alkyne and an allenyne.
-

Definition of pericyclic reactions What is cyclo addition reaction ? Woodward-Hoffmann selection rules for cyclo addition reaction are as follows, Exo-cyclo addition reaction Endo cyclo addition reaction What is Diels Alder reaction ? ( 4+4) type cyclo addition reaction:
-

The "retro-Diels Alder" reaction is the exact reverse of the Diels-Alder and passes through the same transition state. Let's look at some classic examples.
-

The Diels-Alder reaction has long been a powerful tool in organic synthesis. In recent years, the Alder ene reaction has also achieved some prominence. From the beginning, it was apparent that the intramolecular variants of these reactions would be feasible. Many such have been reported, but the results are widely scattered in the chemical literature. This volume is an attempt to synthesize results observed to date, and to suggest directions for future development. One of the limiting factors in the application of the intramolecular Diels- Alder reaction has been the development of methods for the preparation of the requisite trienes. The fIrst chapter of this volume summarizes methods for the preparation of dienes and dienophiles. Examples representative of every general approach to 1,3-dienes and to dienophilic functional group combinations have been included. There are two questions one might ask in considering the prospective cyclization of a given triene: what are the factors that govern the rate of cyclization? and, for cyclizations that lead to the creation of one or more new chiral centers, what are the factors that govern diastereoselectivity? These questions are addressed in Chapter Two. The third chapter is devoted to the all-carbon intramolecular Alder ene reaction. The tables in that chapter summarize all examples that could be found in the literature through 1981, with several additional examples from 1982. Leading references to heterocyclic ene reactions are also included in this chapter.
-

Diels Alder Reaction Organic Chemistry tutorial video for Cyclic Reactants and Bicyclo Products. Learn how to tackle these examples and how to apply my trick for quick results
-
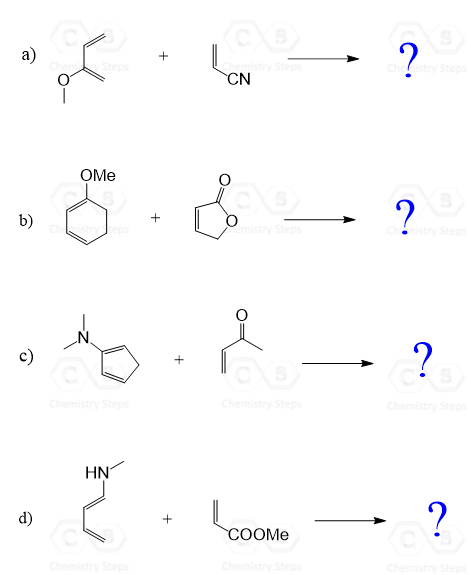
In these practice problems, we will determien the product of Diels-Alder reactions based on the regioselectivitve reaction of the Diene and Dionophile.
-

This book systematically reviews recent advances in the synthetic methods and applications of helicenes. The first part of this book introduces the nomenclature and structural features of helicenes. The second part reviews several classic and useful methods as well as recently-developed approaches for the preparation and functionalization of helicenes,…
-

When non-symmetrical dienes react with non-symmetrical dienophiles in the Diels Alder reaction, two regioisomers are possible. Let's look at some examples.

